Scientific Data Surah 83 · Ayah 9
The science behind kindling a firel
The science behind kindling a firel
The science behind kindling a fire
#1
How Fire Works
BY TOM HARRIS
What is Fire?
PREV NEXT

What exactly are those orange flames?
ROLFO ROLF BRENNER/GETTY IMAGES
Typically, fire comes from a chemical reaction between oxygen in the atmosphere and some sort of fuel (wood or gasoline, for example). Of course, wood and gasoline don't spontaneously catch on fire just because they're surrounded by oxygen. For the combustion reaction to happen, you have to heat the fuel to its ignition temperature.
Here's the sequence of events in a typical wood fire:
Something heats the wood to a very high temperature. The heat can come from lots of different things -- a match, focused light, friction, lightning, something else that is already burning...
When the wood reaches about 300 degrees Fahrenheit (150 degrees Celsius), the heat decomposes some of the cellulose material that makes up the wood.
Some of the decomposed material is released as volatile gases. We know these gases as smoke. Smoke is compounds of hydrogen, carbon and oxygen. The rest of the material forms char, which is nearly pure carbon, and ash, which is all of the unburnable minerals in the wood (calcium, potassium, and so on). The char is what you buy when you buy charcoal. Charcoal is wood that has been heated to remove nearly all of the volatile gases and leave behind the carbon. That is why a charcoal fire burns with no smoke.
The actual burning of wood then happens in two separate reactions:


• When the volatile gases are hot enough (about 500 degrees F (260 degrees C) for wood), the compound molecules break apart, and the atoms recombine with the oxygen to form water, carbon dioxide and other products. In other words, they burn.
• The carbon in the char combines with oxygen as well, and this is a much slower reaction. That is why charcoal in a BBQ can stay hot for a long time.
A side effect of these chemical reactions is a lot of heat. The fact that the chemical reactions in a fire generate a lot of new heat is what sustains the fire. Many fuels burn in one step. Gasoline is a good example. Heat vaporizes gasoline and it all burns as a volatile gas. There is no char. Humans have also learned how to meter out the fuel and control a fire. A candle is a tool for slowly vaporizing and burning wax.
As they heat up, the rising carbon atoms (as well as atoms of other material) emit light. This "heat produces light" effect is called incandescence, and it is the same kind of thing that creates light in a light bulb. It is what causes the visible flame. Flame color varies depending on what you're burning and how hot it is. Color variation within in a flame is caused by uneven temperature. Typically, the hottest part of a flame -- the base -- glows blue, and the cooler parts at the top glow orange or yellow.
In addition to emitting light, the rising carbon particles may collect on surrounding surfaces as soot.


Fire forms a sphere in microgravity.
PHOTO COURTESY NASA
The dangerous thing about the chemical reactions in fire is the fact that they are self-perpetuating. The heat of the flame itself keeps the fuel at the ignition temperature, so it continues to burn as long as there is fuel and oxygen around it. The flame heats any surrounding fuel so it releases gases as well. When the flame ignites the gases, the fire spreads.
On Earth, gravity determines how the flame burns. All the hot gases in the flame are much hotter (and less dense) than the surrounding air, so they move upward toward lower pressure. This is why fire typically spreads upward, and it's also why flames are always "pointed" at the top. If you were to light a fire in a microgravity environment, say onboard the space shuttle, it would form a sphere!
Fire Variables
PREV UP NEXT
In the last section, we saw that fire is the result of a chemical reaction between two gases, typically oxygen and a fuel gas. The fuel gas is created by heat. In other words, with heat providing the necessary energy, atoms in one gaseous compound break their bonds with each other and recombine with available oxygen atoms in the air to form new compounds plus lots more heat.
Only some compounds will readily break apart and recombine in this way -- the various atoms have to be attracted to each other in the right manner. For example, when you boil water, it takes the gaseous form of steam, but this gas doesn't react with oxygen in the air. There isn't a strong enough attraction between the two hydrogen atoms and one oxygen atom in a water molecule and the two oxygen atoms in an oxygen molecule, so the water compound doesn't break apart and recombine.
The most flammable compounds contain carbon and hydrogen, which recombine with oxygen relatively easily to form carbon dioxide, water and other gases.
Different flammable fuels catch fire at different temperatures. It takes a certain amount of heat energy to change any particular material into a gas, and even more heat energy to trigger the reaction with oxygen. The necessary heat level varies depending on the nature of the molecules that make up the fuel. A fuel's piloted ignition temperature is the heat level required to form a gas that will ignite when exposed to a spark. At the unpiloted ignition temperature, which is much higher, the fuel ignites without a spark.
The fuel's size also affects how easily it will catch fire. A larger fuel, such as a thick tree, can absorb a lot of heat, so it takes
Link: https://science.howstuffworks.com/environmental/earth/geophysics/fire1.htm
#2
How and why fires burn
The secret of fire: Prometheus to pyrolysis

On Earth’s surface, gravity gives flames their characteristic shape (right). But in the weak gravity of space, a flame appears as a sphere (left).
Some ancient Greeks believed that fire was a basic element of the universe — one that gave rise to other elements, like earth, water and air. (Aether, that stuff the ancients thought stars were made of, was later added to the list of elements by the philosopher Aristotle.)
Now scientists use the word “element” to describe the most basic types of matter. Fire does not qualify.
A fire’s colorful flame results from a chemical reaction known as combustion. During combustion, atoms rearrange themselves irreversibly. In other words, when something burns, there’s no un-burning it.
Fire also is a glowing reminder of the oxygen that pervades our world. Any flame requires three ingredients: oxygen, fuel and heat. Lacking even one, a fire won’t burn. As an ingredient of air, oxygen is usually the easiest to find. (On planets such as Venus and Mars, with atmospheres containing far less oxygen, fires would be hard to start.) Oxygen’s role is to combine with the fuel.
Any number of sources may supply heat. When lighting a match, friction between the match’s head and the surface against which it’s struck releases enough heat to ignite the coated head. In the Avalanche Fire, lightning delivered the heat.
Fuel is what burns. Almost anything can burn, but some fuels have a far higher flash point — the temperature at which they’ll ignite — than others.
People feel heat as warmth on the skin. Not atoms. The building blocks of all materials, atoms just get antsy as they warm. They initially vibrate. Then, as they warm even more, they start dancing, faster and faster. Apply enough heat, and atoms will break the bonds linking them together.
Wood, for example, contains molecules made from bound atoms of carbon, hydrogen and oxygen (and smaller amounts of other elements). When wood gets hot enough — such as when lightning hits or a log is tossed on an already burning fire — those bonds break. The process, called pyrolysis, releases atoms and energy.
Unbound atoms form a hot gas, mingling with oxygen atoms in the air. This glowing gas — and not the fuel itself — produces the spooky blue light that appears at the base of a flame.
But the atoms don’t stay single long: They quickly bond with oxygen in the air in a process called oxidation. When carbon bonds with oxygen, it produces carbon dioxide — a colorless gas. When hydrogen bonds with oxygen, it produces water vapor — even as the wood burns.
Fires burn only when all that atomic shuffling releases enough energy to keep the oxidation going in a sustained chain reaction. More atoms released from the fuel combine with nearby oxygen. That releases more energy, which releases more atoms. This heats the oxygen — and so on.
The orange and yellow colors in a flame appear when extra, free-floating carbon atoms get hot and begin to glow. (These carbon atoms also make up the thick black soot that forms on grilled burgers or the bottom of a pot heated over a fire.)
Power Words
atom The basic unit of a chemical element.
bond (in chemistry) Attractive forces between atoms that are strong enough to make the linked elements function as a single unit. Some of the attractive forces are weak, some are very strong. All bonds appear to link atoms through a sharing of — or an attempt to share — electrons.
carbon The chemical element having the atomic number 6. It is the physical basis of all life on Earth. Carbon exists freely as graphite and diamond. It is an important part of coal, limestone and petroleum, and is capable of self-bonding, chemically, to form an enormous number of chemically, biologically and commercially important molecules.
chemical reaction A process that involves rearrangement of the molecules or structure of a substance, as opposed to a change in physical form.
climate The weather conditions prevailing in an area in general or over a long period.
combust To consume by fire. Combustion is the process of burning.
element Each of more than one hundred substances that cannot be broken down into simpler substances.
fire The burning of some fuel, creating a flame that releases light and heat.
fuel Any material that will release energy during a controlled chemical or nuclear reaction. Fossil fuels (coal, natural gas and petroleum) represent a common type that liberates its energy through chemical reactions that take place when heated (usually to the point of burning).
molecule An electrically neutral group of atoms that represents the smallest possible amount of a chemical compound. Molecules can be made of single types of atoms or of different types. For example, the oxygen in the air is made of two oxygen atoms (O2), but water is made of two hydrogen atoms and one oxygen atom (H2O).
oxygen A gas that makes up about 21 percent of the atmosphere. All animals and many microorganisms need oxygen to fuel their metabolism.
pyrolysis A chemical change triggered by heat, often due to fire.
Link: https://www.sciencenewsforstudents.org/article/explainer-how-and-why-fires-burn
#3
Fire behaviour
Fires behave differently. Some burn slowly and evenly; others are extremely hot, burning fiercely and quickly. Different fires have different coloured flames. Some fires start easily; others don’t. Some fires produce deadly gases that could kill you if not ventilated.

Log fire
Fires are different depending on the variables involved. This log fire is a controlled fire, designed to radiate heat.
The behaviour of the fire often depends on the fuel. Other factors or variables may include where the fuel is situated and how near it is to other fuels, the weather (especially wind and relative humidity), oxygenconcentration and, in the case of outside fires, the shape of the terrain.
Structural fires
In this video clip, Associate Professor Charley Fleischmann explains what burns in structural fires are and what makes them so dangerous.
Fuel
Type: Different fuels catch fire at different temperatures. It takes a certain amount of heat energy to change any particular material into a gas (if it is not already). Then it takes more heat energy to trigger the reaction with oxygen. The amount of heat produced depends on the molecules that make up the fuel. The most flammable fuels are hydrocarbons (contain carbon and hydrogen) that recombine with oxygen quite easily to form carbon dioxide, water and other gases.

Wildfire
Fires are different depending on the variables involved. A wildfire is uncontrolled, often intense and spreads rapidly.
Size: How quickly a fuel catches fire and burns relates to the surface area or the size of the fuel. For example, large pieces of wood take a lot longer to absorb heat energy to ignition temperature. A twig catches fire easily because it heats up easily.
Surface area: The bigger the area of the surface of the fuel, the more oxygen molecules can collide with the surface. The more oxygen molecules that collide per second with the fuel, the faster the combustionreaction is.
You can increase the surface area of a solid by breaking it up into smaller pieces. If you chop or break up wood into small pieces, it will ignite and burn more quickly than larger pieces of wood. People often start fires with kindling (small pieces of wood) that they criss-cross to allow greater surface area and lots of oxygen getting in and around.
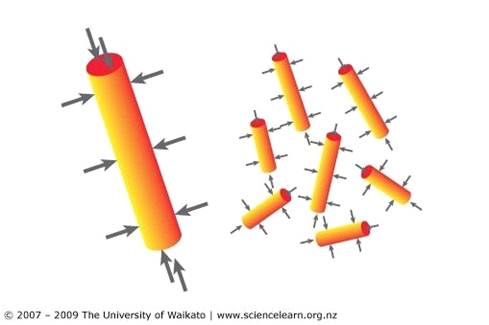
Differences in surface area
You can increase the surface area of a solid by breaking it up into smaller pieces. If you chop up wood into small pieces, it will ignite and burn more quickly than larger pieces of wood.
A powder has the largest surface area and will have the fastest reaction rate.
Heat produced: How much energy is released in the reaction and how quickly the fuel burns depends on what the fuel is made up of. Different compounds react with oxygen differently – some contain lots of heat energy while others produce a smaller amount. The reaction with the oxygen may happen very quickly or more slowly.
Amount: The amount of fuel available to burn is known as the fuel load. The bigger the fuel load, the more intense the fire will be in terms of heat energy output.
Moisture content: If the fuel isn’t dry enough, it won’t burn. The less moisture in the fuel, the more likely it will ignite and burn.
Oxygen availability
The amount of oxygen available will affect the rate of burning. A low concentration of oxygen will slow the burning right down.
An example of dangerous fire behaviour that can occur in a situation where there is a low concentration of oxygen is called backdraught. This is when an enclosed fire has used up most of the oxygen and is just smouldering. If there is a sudden influx of oxygen (like someone opening a door or window), the fire will immediately explode into flame.
Flashover and backdraught
In this video clip, Associate Professor Charley Fleischmann describes how flashover and backdraught occur in fires.
Weather
Relative humidity: This reflects the amount of moisture in the air. If relative humidity is low, it will contribute to the drying of fuels. If it is high, fuels will absorb moisture from the air, making ignition more difficult.
Wind: This is a major factor in determining fire spread. Wind affects the rate of oxygen supply to the burning fuel (controlling combustion) and it tilts the flame forward so that unburned fuel receives energy by radiation and convection at an increased speed. Wind can also dry out the fuel.
Rainfall: This also has an effect on wetting fuels, but absorption of moisture is dependent on fuel size. Fine fuels absorb moisture more quickly than coarse fuels. Lack of rain (precipitation) is the biggest factor affecting the drying process of fuels.
Increased temperatures: These will dry out potential fuel so that there will be less preheating of fuels to reach ignition temperature.
Terrain
The terrain (shape of the land) has significant influence on wildfire behaviour. Steep slopes may increase fire speed because fuels (scrub and vegetation) are preheated ahead of the fire through convection and radiation.
Rural fires
In this video, Stuart Anderson talks about rural fires compared to structural fires.
Rugged terrain with narrow valleys, sharp ridges and irregular slopes affect the direction and rate of fire spread. For example, narrow valleys can funnel winds, increasing the rate of spread of a fire due to convection.
The direction a slope is facing will depend on how much sun it gets. This will affect the amount of drying the fuels get. The drier the fuel, the faster it will burn.
Link: https://www.sciencelearn.org.nz/resources/763-fire-behaviour